Content of the box
- 20 Tests individually foiled (Cassettes)
- Buffer
- Sample prep kit & swabs
- Package Insert (Instructions)
INTENDED USE
The Coronavirus Ag Rapid Test Cassette (Swab) is an in vitro immunochromatographic assay for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2 in direct nasal swab specimens directly from individuals who are both symptomatic and asymptomatic. It is intended to aid in the rapid diagnosis of SARSCoV-2 infections. Negative results from patients with symptom onset beyond ten days, should be treated as presumptive and confirmation with a molecular assay, if necessary, for patient management, may be performed.
The Coronavirus Ag Rapid Test Cassette (Swab) does not differentiate between SARS-CoV and SARS-CoV-2. Coronavirus Ag Rapid Test Cassette (Swab) is intended for use by healthcare professionals or other trained operators who are proficient in performing rapid tests. Equally, healthcare professionals & trained operators can oversee non-trained personnel to undertake all steps of the test themselves as long as its carried out in a supervised capacity.
SUMMARY AND EXPLANATION
The novel coronaviruses belong to the β genus. COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue, and dry cough. Nasal congestion, runny nose, sore throat, myalgia, and diarrhea are found in a few cases. This test is for detection of SARS-CoV-2 nucleocapsid protein antigen. Antigen is generally detectable in upper respiratory specimens during the acute phase of infection. Rapid diagnosis of SARS-CoV-2 infection will help healthcare professionals to treat patients and control the disease more efficiently and effectively. To effectively monitor the SARS-CoV-2 pandemic, systematic screening and detection of both clinical and asymptomatic COVID-19 cases is critical. Particularly, the identification of subclinical or asymptomatic cases is
important to reduce or stop the infection because these individuals may transmit the virus. Coronavirus Ag Rapid Test Cassette (Swab) allows effective screening of COVID-19 infection.
PRINCIPLE OF THE TEST
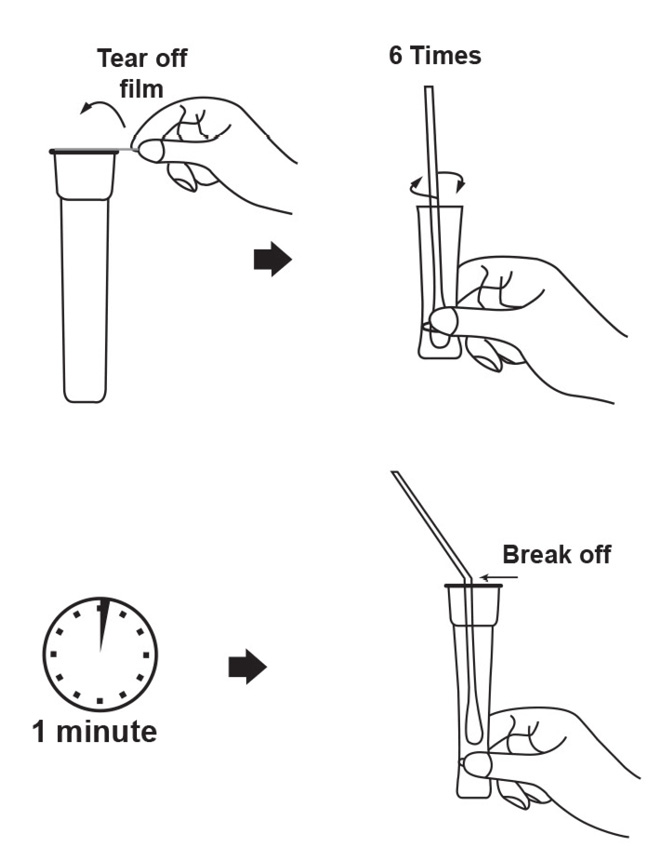
The Coronavirus Ag Rapid Test Cassette (Swab) is an immunochromatographic membrane assay that uses highly sensitive monoclonal antibodies to detect nucleocapsid protein from SARS-CoV-2 in direct nasal swab. The test strip is composed of the following parts: namely sample pad, reagent pad, reaction membrane, and absorbing pad.
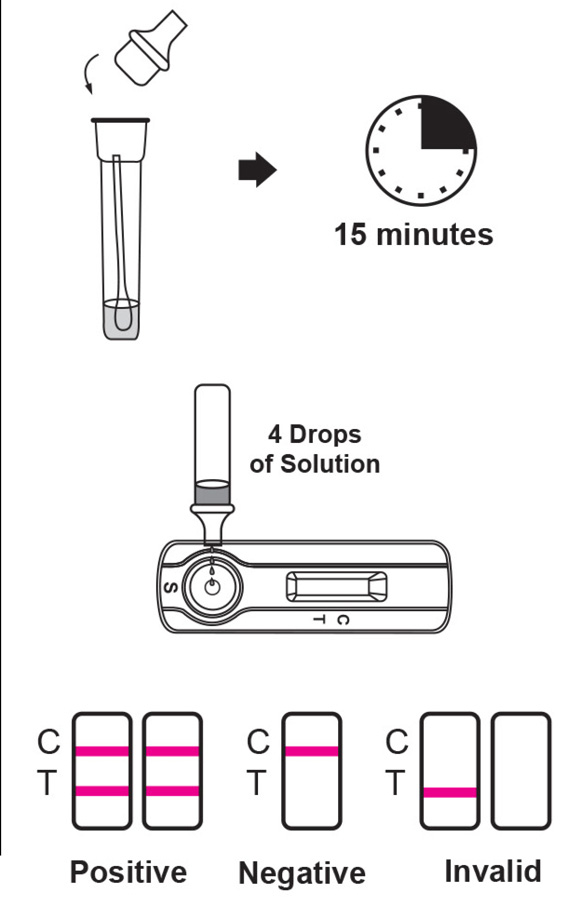
The reagent pad contains the colloidal-gold conjugated with the monoclonal antibodies against the nucleocapsid protein of SARS-CoV-2; the reaction membrane contains the secondary antibodies for nucleocapsid protein of SARS-CoV-2. The whole strip is fixed inside a plastic device. When the sample is added into the sample well, conjugates dried in the reagent pad are dissolved and migrate along with the sample. If SARS-CoV-2 nucleocapsid antigen is present in the sample, a complex forms between the anti-SARS-2 conjugate and the virus will be captured by the specific anti-SARS-2 monoclonal antibodies coated on the test line region (T). Absence of the test line (T) suggests a negative result. To serve as a procedural control, a red line will always appear in the control line region (C) indicating that proper volume of sample has been added and membrane wicking has occurred.
MATERIALS PROVIDED
1. Test Cassettes)
2. Sterile Nasal Swabs
3. Extraction Tubes with Buffer, Tips and Caps
4. 1/2 Workstation(s)
5. 1 Package Insert
6. Zip Lock PP Bags
MATERIALS REQUIRED BUT NOT PROVIDED
1. Clock, timer, or stopwatch
SAHPRA Manufacturing license Antigen test










